Sunday, 19 May 2013
5.11 understand that, in car engines, the temperature reached is high enough to allow nitrogen and oxygen from air to react, forming nitrogen oxides
The temperature in a car engine is high enough for nitrogen and oxygen from the air to react to form oxides of nitrogen. These oxides are also passed out through the exhaust of the car and when they get into the atmosphere, they can dissolve in the water in the air to form acid rain.
5.10 understand that incomplete combustion of fuels may produce carbon monoxide and explain that carbon monoxide is poisonous because it reduces the capacity of the blood to carry oxygen
Incomplete combustion of an alkane produces carbon monoxide. The same is true when petrol or diesel is burnt in cars, some of the fuel is not completely burnt because of the lack of oxygen.
The carbon monoxide produced passes out through the exhaust pipe of the car and gets into the atmosphere. This is potentially dangerous, since carbon monoxide is poisonous to humans as it reduces the capacity of the blood to carry oxygen.
The carbon monoxide produced passes out through the exhaust pipe of the car and gets into the atmosphere. This is potentially dangerous, since carbon monoxide is poisonous to humans as it reduces the capacity of the blood to carry oxygen.
5.9 describe the trend in boiling point and viscosity of the main fractions
The lower down the fraction, the higher the boiling point.
The higher the boiling point, the lower the viscosity, the lower the boiling point the higher the viscosity.
5.8 recall the names and uses of the main fractions obtained from crude oil: refinery gases, gasoline, kerosene, diesel, fuel oil and bitumen
Uses of the products of the main fractions
Refinery gases: bottles gas for camping etc.
Gasoline: petrol for cars
Kerosene: fuel for aeroplanes
Diesel oil: fuel for buses, lorries, trains and cars
Fuel oil: fuel for ships and industrial heating
Bitumen: road surfaces and covering flat roofs for buildings.
Refinery gases: bottles gas for camping etc.
Gasoline: petrol for cars
Kerosene: fuel for aeroplanes
Diesel oil: fuel for buses, lorries, trains and cars
Fuel oil: fuel for ships and industrial heating
Bitumen: road surfaces and covering flat roofs for buildings.
5.7 describe and explain how the industrial process of fractional distillation separates crude oil into fractions
Fractional distillation
Crude oil, as such, has no direct use. It has to be refined before it is any use. The first step in the refining of crude oil is fractional distillation.
Fractional distillation is carried out in a fractionating column. The column is hot at the bottom and gradually becomes cooler at the top.
The crude oil is split into various fractions as described below. A fraction is a mixture of hydrocarbons with very similar boiling points.
Crude oil is heated to convert it into a vapour. The vapour is then fed into the bottom of the fractionating column.
The hydrocarbons with very high boiling points (fuel, oil and bitumen) immediately turn into liquids and are tapped off at the bottom of the column.

Crude oil, as such, has no direct use. It has to be refined before it is any use. The first step in the refining of crude oil is fractional distillation.
Fractional distillation is carried out in a fractionating column. The column is hot at the bottom and gradually becomes cooler at the top.
The crude oil is split into various fractions as described below. A fraction is a mixture of hydrocarbons with very similar boiling points.
Crude oil is heated to convert it into a vapour. The vapour is then fed into the bottom of the fractionating column.
The hydrocarbons with very high boiling points (fuel, oil and bitumen) immediately turn into liquids and are tapped off at the bottom of the column.

5.6 understand that crude oil is a mixture of hydrocarbons
Crude oil is a thick, sticky, black liquid that is found under the ground and under the sea in certain parts of the world such as he Middle East and Texas, USA.
It is a mixture of hydrocarbons, mostly alkanes.
It is a mixture of hydrocarbons, mostly alkanes.
5.5 explain the uses of aluminium and iron, in terms of their properties.
Uses of aluminium and iron
Aluminium :
Aluminium :
- aeroplane bodies: high strength-to-weight ratio
- overhead power cables: good conductor of electricity
- saucepans: good conductor of heat
- food cans: non-toxic
- window frames: resists corrosion
Iron
- car bodies: strong
- iron nails: strong
- ships, girders and bridges: strong
5.3 write ionic half-equations for the reactions at the electrodes in aluminium extraction
Half equations
The reaction at the negative electrode (cathode) for aluminium extraction is:
Al3- + 3e----> Al
The reaction at the positive electrode (anode) for aluminium extraction is:
2O2- ----> O2 + 4e
The reaction at the negative electrode (cathode) for aluminium extraction is:
Al3- + 3e----> Al
The reaction at the positive electrode (anode) for aluminium extraction is:
2O2- ----> O2 + 4e
5.2 describe and explain the extraction of aluminium from purified aluminium oxide by electrolysis, including: i the use of molten cryolite as a solvent and to decrease the required operating temperature ii the need to replace the positive electrodes iii the cost of the electricity as a major factor

Aluminium oxide has a very high melting point and hence it is dissolved in molten cryolite to make the electrolyte. This mixture has a much lower melting point and is also better conductor of electricity than molten aluminium oxide.
The electrodes are made of graphite (carbon) and they need to be replaced regularly because of the hot temperatures and the carbon anodes burn with oxygen to create carbon dioxide. This means that they need to be replaced regularly and this adds to the cost of the extraction.
Large amounts of energy are needed to produce aluminium, this is why using molten cryolite saves quite a lot of money, as it has a reasonably low melting point and acts as a solvent for aluminium oxide.
5.1 explain how the methods of extraction of the metals in this section are related to their positions in the reactivity series
Methods of extraction
The metal of extraction is linked to the position of the metal in the reactivity series, since the more reactive metals are better at 'keeping hold' if the elements in their compounds. This means the more reactive the metal, the more difficult and expensive it is to extract.
The metal of extraction is linked to the position of the metal in the reactivity series, since the more reactive metals are better at 'keeping hold' if the elements in their compounds. This means the more reactive the metal, the more difficult and expensive it is to extract.
4.25 predict the effects of changing the pressure and temperature on the equilibrium position in reversible reactions.
By changing the pressure and temperature it is possible to shift the position of the equilibrium of the reaction.
4.24 understand the concept of dynamic equilibrium
Dynamic equilibrium
Dynamic equilibrium is when a reversible reaction is happening both ways at the same time, at the same rate.
Dynamic equilibrium is when a reversible reaction is happening both ways at the same time, at the same rate.
4.23 describe reversible reactions such as the dehydration of hydrated copper(II) sulfate and the effect of heat on ammonium chloride
Adding water to copper (II) sulfate, makes it hydrated.
If you remove water from the hydrated copper (II) sulfate, it becomes dehydrated.
4.22 understand that some reactions are reversible and are indicated by the symbol ⇌ in equations
⇌ This symbol represents that a reaction is reversible, which means the reaction occurs in both direction.
4.19 understand the term activation energy and represent it on a reaction profile
Colliding particles must have sufficient energy to react. This energy is known as the activation energy. Collisions that have the required activation energy are called successful collisions.
4.18 describe the effects of changes in surface area of a solid, concentration of solutions, pressure of gases, temperature and the use of a catalyst on the rate of a reaction
Increasing the concentration of a reactant increases the number of particles of reactant in a given volume and hence the reacting particles will collide more often. Hence, there will be more successful collisions per second.
Increasing the temperature increases the average kinetic energy of the reactant particles and therefore more of the collisions that take place will have the necessary activation energy to react, hence there will be more successful collisions per second.
Increasing the surface area means that more particles become in contact with other particles of the reactant. Particles collide more often, therefore more successful collisions per second.
Adding a catalysts increases the rate of reaction because it slows down the reaction, giving it 'a lower activation energy' than the original speed. More collisions will be taking place, there will be more successful collisions per second.
Increasing the temperature increases the average kinetic energy of the reactant particles and therefore more of the collisions that take place will have the necessary activation energy to react, hence there will be more successful collisions per second.
Increasing the surface area means that more particles become in contact with other particles of the reactant. Particles collide more often, therefore more successful collisions per second.
Adding a catalysts increases the rate of reaction because it slows down the reaction, giving it 'a lower activation energy' than the original speed. More collisions will be taking place, there will be more successful collisions per second.
4.17 describe experiments to investigate the effects of changes in surface area of a solid, concentration of solutions, temperature and the use of a catalyst on the rate of a reaction
Factors that effect the rate of a reaction
Surface area
Concentration
Temperature
Catalyst
Surface area
- Put a set mass of magnesium in hydrochloric acid
- Time the reaction
- Change the from of magnesium keeping the mass the same (powder, wire, strips)
- The more surface area (the smaller the pieces of magnesium) the faster the reaction
Concentration
- Put a set mass of marble chips into dilute hydrochloric acid
- Time the reaction
- Change the ratio of water to hydrochloric acid
- The more concentrated the hydrochloric acid (the lower the ratio of water) the faster the reaction
Temperature
- Put a set mass of magnesium powder into a set mass of hydrochloric acid
- time the reaction
- Carry out this reaction at different temperatures
- The higher the temperature the faster the rate of reaction
Catalyst
- If you have hydrogen peroxide it will not decompose
- If you put it with manganese dioxide it will decompose into water and oxygen
- The manganese dioxide will be unaltered by the reaction
- The more of the catalyst the faster the reaction
TAKEN FROM HANNAHHELPCHEMISTRY
4.15 understand that the breaking of bonds is endothermic and that the making of bonds is exothermic
The energy required to break a bond is known as the bond energy. It is usually measured in kilojoules per mole of bonds (kK/mol).
Breaking bonds takes in energy (endothermic)
Creating bonds gives out energy (exothermic)
Breaking bonds takes in energy (endothermic)
Creating bonds gives out energy (exothermic)
4.13 understand the use of ΔH to represent enthalpy change for exothermic and endothermic reactions
The amount of thermal energy change is given the symbol ΔH.
For an exothermic reaction, ΔH is given a negative sign.
For an endothermic reaction, ΔH is given a positive sign.
For an exothermic reaction, ΔH is given a negative sign.
For an endothermic reaction, ΔH is given a positive sign.
4.11 describe simple calorimetry experiments for reactions such as combustion, displacement, dissolving and neutralisation in which heat energy changes can be calculated from measured temperature changes
Record a solution's temperature in the beginning of an experiment and at the end of the experiment, what ever the temperature difference is, is the calorimetry of that experiment.
4.10 understand that chemical reactions in which heat energy is given out are described as exothermic and those in which heat energy is taken in are endothermic
Exothermic reaction: thermal energy is given out.
Endothermic reaction: thermal energy is taken in.
Endothermic reaction: thermal energy is taken in.
4.9 describe experiments to carry out acid-alkali titrations.
Acid-alkali titrations
A titration is a method of finding out exactly the volume of one solution that is required to react with a given volume of another solution.
Titrations are commonly used to find out the volume of acid required to react exactly with a given volume of an alkali:
A titration is a method of finding out exactly the volume of one solution that is required to react with a given volume of another solution.
Titrations are commonly used to find out the volume of acid required to react exactly with a given volume of an alkali:
- Using a pipette, put 25.0cm3 of an alkali solution into a conical flask.
- Add a few drops of an indicator, such as methyl orange.
- Put the acid in a pipette and note the initial reading.
- Add the acid to the alkali until the indicator just changes colour.
- Not the final reading of acid in the burette.
- Subtract the initial reading to obtain the volume of acid added. This is the volume required to neutralise the 25.0cm3 of the alkali.
4.8 describe experiments to prepare insoluble salts using precipitation reactions
Preparation of insoluble salts
Insoluble salts are made by precipitation reactions.
A precipitate is an insoluble solid that is made by a chemical reaction that takes place in a aqueous solution.
A precipitation reaction is a reaction that produces a precipitate.
To make an insoluble salt, it is necessary to mix together two separate aqueous solutions. One solution must contain the required positive ion and the other solution the required negative ion.
The precipitate is then removed by filtration, washed with a little distilled water and left to dry in a warm place.
Insoluble salts are made by precipitation reactions.
A precipitate is an insoluble solid that is made by a chemical reaction that takes place in a aqueous solution.
A precipitation reaction is a reaction that produces a precipitate.
To make an insoluble salt, it is necessary to mix together two separate aqueous solutions. One solution must contain the required positive ion and the other solution the required negative ion.
The precipitate is then removed by filtration, washed with a little distilled water and left to dry in a warm place.
4.7 describe experiments to prepare soluble salts from acids
Preparing soluble salts from acids
Acid + insoluble base
Acid + insoluble base
- Put some dilute acid into a beaker and heat it using a Bunsen burner flame. Do not let it boil.
- Add the insoluble base, a little at a time, to the warm dilute acid and stir until the base is in excess.
- Filter the mixture into an evaporating basin to remove the excess base.
- Leave the filtrate in a warm place so the water evaporates and crystals form.
- Remove the crystals and dry them on filter paper.
- Put an aqueous solution of the alkali into a conical flask and add a suitable indicator (e.g. litmus or methyl orange)
- Add dilute acid from a burette until the indicator just changes colour.
- Add powered charcoal and shake the mixture to remove the colour of the indicator.
- Filter to remove the charcoal and then obtain crystals from the filtrate in the usual manner.
4.6 understand the general rules for predicting the solubility of salts in water: i all common sodium, potassium and ammonium salts are soluble ii all nitrates are soluble iii common chlorides are soluble, except silver chloride iv common sulfates are soluble, except those of barium and calcium v common carbonates are insoluble, except those of sodium, potassium and ammonium
General rules of solubility
Soluble salts
Insoluble salts
Soluble salts
- all common sodium, potassium and ammonium salts
- all nitrates
- all common chlorides (except silver and lead (II) chloride)
- all common sulfates (except barium,calcium and lead (II) sulfate)
- sodium, potassium and ammonium carbonates
Insoluble salts
- silver chloride and lead (II) chloride
- barium sulfate, calcium sulfate and lead (II) sulfate
- all other common carbonates
Saturday, 18 May 2013
4.4 define acids as sources of hydrogen ions, H+ , and alkalis as sources of hydroxide ions, OH¯
Acids and alkalis
An acid (at GCSE) is a substance that dissolves in water to produce hydrogen ions (H+)
An alkali is a substance that dissolves in water to produce hydroxide ions (OH-)
An acid (at GCSE) is a substance that dissolves in water to produce hydrogen ions (H+)
An alkali is a substance that dissolves in water to produce hydroxide ions (OH-)
4.3 describe the use of universal indicator to measure the approximate pH value of a solution
Universal indicator can come in two forms, either in a liquid solution of ethanol or in a paper that has been soaked in the indicator solution.
Either drop some universal indicator into the solution or drop the liquid under test onto the paper.
Either drop some universal indicator into the solution or drop the liquid under test onto the paper.
4.1 describe the use of the indicators litmus, phenolphthalein and methyl orange to distinguish between acidic and alkaline solutions
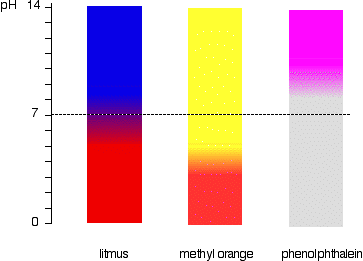
0-6 is acid
8-14 is alkali
7 is neutral
3.8 describe the addition reaction of alkenes with bromine, including the decolourising of bromine water as a test for alkenes.
Alkenes undergo addition reactions with halogens. For example, a bromine molecule will add across the double bond of ethene to form 1,2-dibromoethane.


Since all alkenes contain a carbon- carbon double bond, they will all decolourise bromine.
This reaction, therefore can be used to test for the presence of a carbon-carbon double bond.
Saturated compounds will not immediately decolourise bromine.
3.6 recall that alkenes have the general formula Cn H2n
Alkenes
The alkenes are another homologous series of compounds that have the general formila CnH2n
The alkenes are another homologous series of compounds that have the general formila CnH2n
3.5 describe the substitution reaction of methane with bromine to form bromomethane in the presence of UV light.
Methane and bromine react together in the presence of ultra-violet radiation to form bromomethane.
CH4 (g) + Br2 (g) ---> CH3 Br(g) + HBr (g)
Since one atom in methane (a hydrogen atom) has been replaced by another atom (a bromine atom) this reaction is called a substitution reaction.
CH4 (g) + Br2 (g) ---> CH3 Br(g) + HBr (g)
Since one atom in methane (a hydrogen atom) has been replaced by another atom (a bromine atom) this reaction is called a substitution reaction.
3.2 recall that alkanes have the general formula Cn H2n+2
Alkane
The alkanes are a homologous series of compounds that have the general formula of CnH2n+2
The alkanes are a homologous series of compounds that have the general formula of CnH2n+2
3.1 explain the terms homologous series, hydrocarbon, saturated, unsaturated, general formula and isomerism.
Organic chemistry introduction
homologous series: a series of organic compounds that have the same general formula, similar chemical reactions and where each member differ from the next by a given formula.
hydrocarbon: a compound only containing carbon and hydrogen
saturated: an organic compound in which all the bonds are single bonds
unsaturated: an organic compound that contains a carbon- carbon double bond
general formula: a formula that states the ratio of atoms of each element in the formula of every compound in a particular homologous series
isomerism: compounds that have the same molecular formula but different formulae are said to exhibit isomerism, the different compounds are called isomers
homologous series: a series of organic compounds that have the same general formula, similar chemical reactions and where each member differ from the next by a given formula.
hydrocarbon: a compound only containing carbon and hydrogen
saturated: an organic compound in which all the bonds are single bonds
unsaturated: an organic compound that contains a carbon- carbon double bond
general formula: a formula that states the ratio of atoms of each element in the formula of every compound in a particular homologous series
isomerism: compounds that have the same molecular formula but different formulae are said to exhibit isomerism, the different compounds are called isomers
2.39 describe tests for the gases: i hydrogen ii oxygen iii carbon dioxide iv ammonia v chlorine.
Hydrogen: Mix with air and ignite- 'squeaky pop'
Oxygen: Insert glowing splint- splint relights
Carbon dioxide: Bubble through limewater- limewater turns milky
Ammonia: Damp red litmus paper- turns blue
Chlorine: Damp litmus paper- turns white
Oxygen: Insert glowing splint- splint relights
Carbon dioxide: Bubble through limewater- limewater turns milky
Ammonia: Damp red litmus paper- turns blue
Chlorine: Damp litmus paper- turns white
2.38 describe tests for the anions: i Cl- , Br- and I- , using dilute nitric acid and silver nitrate solution ii SO4 2- , using dilute hydrochloric acid and barium chloride solution iii CO3 2- , using dilute hydrochloric acid and identifying the carbon dioxide evolved
Identification of anions
Test: To an aqueous solution of the solid under test, add some dilute nitric acid followed by a few drops of silver nitrate solution.
chloride ion: white precipitate
bromide ion: cream precipitate
iodide ion: yellow precipitate
Sulfate ions
Test: Add dilute hydrochloric acid followed by a few drops of barium chloride solution with the sulfate ions
Result: White precipitate (of barium sulfate)
Carbonate ions
Test: To either the solid, or an aqueous solution of the solid, under test add dilute hydrochloric (or nitric) acid
Result: Bubbles of gas. Will turn limewater milky.
2.37 describe tests for the cations: i Li+ , Na+ , K+ , Ca2+ using flame tests ii NH4 + , using sodium hydroxide solution and identifying the ammonia evolved iii Cu2+, Fe2+ and Fe3+, using sodium hydroxide solution
Flame tests
Lithium: Red
Potassium: Lilac
Calcium: Brick Red
Sodium: Yellow/orange
Identifying the ammonium ion
Test: Add aqueous sodium hydroxide to the solid, or solution, under test and warm the mixture.
Result: If ammonium ions are present then a pungent-smelling gas is produced. The gas is produced turns damp red litmus paper blue.
Identifying metal cations
Copper (II) : blue precipitate
Iron (II): green precipitate
Iron (III): brown precipitate
Lithium: Red
Potassium: Lilac
Calcium: Brick Red
Sodium: Yellow/orange
Identifying the ammonium ion
Test: Add aqueous sodium hydroxide to the solid, or solution, under test and warm the mixture.
Result: If ammonium ions are present then a pungent-smelling gas is produced. The gas is produced turns damp red litmus paper blue.
Identifying metal cations
Copper (II) : blue precipitate
Iron (II): green precipitate
Iron (III): brown precipitate
2.36 understand the sacrificial protection of iron in terms of the reactivity series.
Sacrificial protection of iron
Iron can be prevented from rusting by what we know about the reactivity series. Zinc is above iron in the reactivity series, so reacts more readily than iron.
So the iron is coated with zinc to stop rusting.
Iron can be prevented from rusting by what we know about the reactivity series. Zinc is above iron in the reactivity series, so reacts more readily than iron.
So the iron is coated with zinc to stop rusting.
2.35 describe how the rusting of iron may be prevented by grease, oil, paint, plastic and galvanising
Preventing rusting
Coating iron with:
Coating iron with:
- grease
- oil
- paint
- plastic
- a metal less reactive than iron, such as tin
2.34 describe the conditions under which iron rusts
Rusting of iron
Rusting is a chemical reaction between iron, water and oxygen. Rusting takes place faster if there are also electrolytes such as sodium chloride in the water. NaCl is present in sea water.
Iron atoms have been oxidised as they have lost 3 electrons.
Rusting is a chemical reaction between iron, water and oxygen. Rusting takes place faster if there are also electrolytes such as sodium chloride in the water. NaCl is present in sea water.
Iron atoms have been oxidised as they have lost 3 electrons.
2.33 understand the terms redox, oxidising agent, reducing agent
Redox reaction- A reaction in which both reduction and oxidation are taking place.
Oxidising agent- A substance in that is capable of oxidising another substance.
Reducing agent- A substance in that is capable of reducing another substance.
Oxidising agent- A substance in that is capable of oxidising another substance.
Reducing agent- A substance in that is capable of reducing another substance.
2.32 understand oxidation and reduction as the addition and removal of oxygen respectively
Oxidation means a gain of oxygen and reduction is a loss of oxygen.
2.31 deduce the position of a metal within the reactivity series using displacement reactions between metals and their oxides, and between metals and their salts in aqueous solutions
If you set up an experiment with a metal oxide or a metal salt
dissolved in water
introduce a more reactive metal and it will displace the current one
introduce a less reactive metal and no displacement will take place
From this you can deduce which metals are more and less reactive.
2.30 describe how reactions with water and dilute acids can be used to deduce the following order of reactivity: potassium, sodium, lithium, calcium, magnesium, zinc, iron and coppe
The higher up the metal, the more vigorously it will react with other elements.
2.29 understand that metals can be arranged in a reactivity series based on the reactions of the metals and their compounds: potassium, sodium, lithium, calcium, magnesium, aluminium, zinc, iron, copper, silver and gold
Reactivity series
Potassium
Sodium
Lithium
Calcium
Magnesium
Aluminium
Zinc
Iron
Tin
Lead
Copper
Silver
Gold
Platinum
The elements at the top are the most reactive and the elements at the bottom are the least reactive.
Potassium
Sodium
Lithium
Calcium
Magnesium
Aluminium
Zinc
Iron
Tin
Lead
Copper
Silver
Gold
Platinum
The elements at the top are the most reactive and the elements at the bottom are the least reactive.
2.28 describe a physical test to show whether water is pure.
Physical test to show whether water is pure
Boil the liquid and measure its boiling point.
If it is water, its boiling point will be 100'C.
Boil the liquid and measure its boiling point.
If it is water, its boiling point will be 100'C.
2.27 describe the use of anhydrous copper(II) sulfate in the chemical test for water
Using anhydrous copper (II) sulfate to test for water
Add the liquid that you are testing to anhydrous copper (II) sulfate. The white powder will turn blue if there is water present in the liquid.
Add the liquid that you are testing to anhydrous copper (II) sulfate. The white powder will turn blue if there is water present in the liquid.
2.26 describe the combustion of hydrogen
Combustion of hydrogen
Hydrogen burns when heated in the air or oxygen to form water.
The product will be formed initially as water vapour, but if cooled it can be condensed to form water.
Hydrogen burns when heated in the air or oxygen to form water.
The product will be formed initially as water vapour, but if cooled it can be condensed to form water.
2.25 describe the reactions of dilute hydrochloric and dilute sulfuric acids with magnesium, aluminium, zinc and iron
Magnesium-
- bubbles of gas
- magnesium disappears
- reaction mixture gets hot
- colourless solution is formed
Aluminium
- slow to start reacting when cold, but bubbles form when heated
- aluminum disappears
- colourless solution formed
Zinc
- bubbles of gas
- zinc disappears
- colourless solution formed
Iron
- bubbles of gas
- iron disappears
- pale green solution formed
2.24 understand that carbon dioxide is a greenhouse gas and may contribute to climate change.
Carbon dioxide is a greenhouse gas since it absorbs infra-red radiation given off from Earth's surface. In recent years the amount of carbon dioxide in Earth's atmosphere has increased. This is because we're burning more fossil fuels than before. Some scientists think that this is contributing to climate change.
2.23 explain the use of carbon dioxide in carbonating drinks and in fire extinguishers, in terms of its solubility and density
Uses of carbon dioxide
- Making carbonated drinks. Even though carbon dioxide is not very soluble in water in normal pressures, it becomes much more soluble when it is put under high pressures. This is a method used to make fizzy drinks.
- In fire extinguishers. Carbon dioxide does not support combustion and is more dense than air, so it 'sits' on top of the burning fuel and prevents oxygen from getting to it.
2.22 describe the properties of carbon dioxide, limited to its solubility and density
Carbon dioxide is more dense than air and is soluble in water at a high pressure.
2.21 describe the formation of carbon dioxide from the thermal decomposition of metal carbonates such as copper(II) carbonate
When green copper (II) carbonate is heated it turns black, and the carbon dioxide and the copper oxide become separated.
2.20 describe the laboratory preparation of carbon dioxide from calcium carbonate and dilute hydrochloric acid
The reaction between any metal carbonate and an acid will form carbon dioxide. Calcium carbonate is the most commonly used metal carbonate in the lab to produce carbon dioxide. The most convenient form of calcium carbonate is marble chips. They are easy to handle and the reaction is not too fast, so the carbon dioxide is produced at a rate that makes it easy to collect.


2.19 describe the reactions of magnesium, carbon and sulfur with oxygen in air, and the acid-base character of the oxides produced
Oxygen is a reactive gas.
Any substance that burns in air will burn more brightly and more vigorously with oxygen.
Magnesium burns with bright, white flame to form a white powder.
Carbon burns with a yellow-orange flame to form a colourless gas, CO2.
Sulfur burns with a blue flame to form a colourless gas.
Any substance that burns in air will burn more brightly and more vigorously with oxygen.
Magnesium burns with bright, white flame to form a white powder.
Carbon burns with a yellow-orange flame to form a colourless gas, CO2.
Sulfur burns with a blue flame to form a colourless gas.
2.18 describe the laboratory preparation of oxygen from hydrogen peroxide, using manganese(IV) oxide as a catalyst
Preparation of oxygen
Hydrogen peroxide, decomposes slowly to form water and oxygen. The speed of the decomposition is increased by adding solid manganese dioxide, which acts as a catalyst.
The oxygen can also be collected over water. Since oxygen is not very soluble in water, very little is lost.
Hydrogen peroxide, decomposes slowly to form water and oxygen. The speed of the decomposition is increased by adding solid manganese dioxide, which acts as a catalyst.
The oxygen can also be collected over water. Since oxygen is not very soluble in water, very little is lost.
2.17 explain how experiments involving the reactions of elements such as copper, iron and phosphorus with air can be used to investigate the percentage by volume of oxygen in air
Using copper:

Set up the apparatus with 100cm3 of air in one of the gas syringes.
Heat the copper at one end of the silica tube using a blue bunsen flame.
Pass the air backwards and forwards over the copper
As the volume of gas in the syringe decreases, move the Bunsen flame along the tube so that it is always heating fresh copper.
Stop heating when the volume of gas in the syringe stops decreasing.
The copper is reacting with the oxygen in the air to form black copper oxide.
The final volume of the air left in the syringe will be approximately 79cm3, this shows that 21% of it has reacted, so there is 21% of oxygen in the air.

Set up the apparatus with 100cm3 of air in one of the gas syringes.
Heat the copper at one end of the silica tube using a blue bunsen flame.
Pass the air backwards and forwards over the copper
As the volume of gas in the syringe decreases, move the Bunsen flame along the tube so that it is always heating fresh copper.
Stop heating when the volume of gas in the syringe stops decreasing.
The copper is reacting with the oxygen in the air to form black copper oxide.
The final volume of the air left in the syringe will be approximately 79cm3, this shows that 21% of it has reacted, so there is 21% of oxygen in the air.
2.16 recall the gases present in air and their approximate percentage by volume
Gases in the air
Nitrogen 78%
Oxygen 21%
Argon 0.9%
Carbon dioxide 0.04%
Nitrogen 78%
Oxygen 21%
Argon 0.9%
Carbon dioxide 0.04%
2.15 understand these displacement reactions as redox reactions.
In displacement reactions, the halogen molecule is gaining electrons and is therefore being reduced.
The halide ions are losing electrons and are therefore being oxidised.
Since both reduction and oxidation are taking place the reaction is a redox reaction.
The halide ions are losing electrons and are therefore being oxidised.
Since both reduction and oxidation are taking place the reaction is a redox reaction.
2.13 describe the relative reactivities of the elements in Group 7
As we go down the group the reactivity decreases because of the increasing size of the atoms. A halogen atom is able to attract an extra electron into its outermost shell to make eight electrons in total. It is the nucleus of each atom that attracts the extra electron.
As the atoms get bigger, their outer electron shell gets further away from the nucleus and hence the force of attraction for an electron decreases.
Therefore the atom gains an extra electron less readily.
As the atoms get bigger, their outer electron shell gets further away from the nucleus and hence the force of attraction for an electron decreases.
Therefore the atom gains an extra electron less readily.
2.12 explain, in terms of dissociation, why hydrogen chloride is acidic in water but not in methylbenzene
Hydrogen chloride is acidic in water because the molecules ionise when dissolved in water. We can prove this because the effect on blue litmus paper is that it turns red.
However hydrogen chloride in methylbenzene does not ionise and therefore is not acidic.
A solution of hydrogen chloride in water is called hydrochloric acid.
However hydrogen chloride in methylbenzene does not ionise and therefore is not acidic.
A solution of hydrogen chloride in water is called hydrochloric acid.
2.11 understand the difference between hydrogen chloride gas and hydrochloric acid
Hydrogen chloride gas and hydrochloric acid
Hydrogen chloride, HCl, is a colourless gas at room temperature. It dissolves in both water and many organic solvents such as methylbenzene.
Hydrochloric acid is HCl dissolved in water, then the two ions H+ and Cl- become detached, leaving the acidic H+, this is why it is an acid.
Hydrogen chloride, HCl, is a colourless gas at room temperature. It dissolves in both water and many organic solvents such as methylbenzene.
Hydrochloric acid is HCl dissolved in water, then the two ions H+ and Cl- become detached, leaving the acidic H+, this is why it is an acid.
Friday, 17 May 2013
2.10 make predictions about the properties of other halogens in this group
Other halogens
Reactivity decreases as we go down the group because of the increasing size of the atoms. A halogen atom is able to attract an extra electron into its outermost shell to make eight electrons in total. It is the nucleus of each atom that attracts the extra electron.
Reactivity decreases as we go down the group because of the increasing size of the atoms. A halogen atom is able to attract an extra electron into its outermost shell to make eight electrons in total. It is the nucleus of each atom that attracts the extra electron.
2.9 recall the colours and physical states of the elements at room temperature
Group 7 elements
Chlorine is a gas at room temperature and is pale green.
Bromine is a liquid at room temperature and is red-brown.
Iodine is a solid at room temperature and is black.
Chlorine is a gas at room temperature and is pale green.
Bromine is a liquid at room temperature and is red-brown.
Iodine is a solid at room temperature and is black.
TRIPLE: 2.8 explain the relative reactivities of the elements in Group 1 in terms of distance between the outer electrons and the nucleus.
Outer electrons of Group 1
The electronic configuration will show the outer electrons.
Li 2.1
Na 2.8.1
K 2.8.8.1
The outer shell has only one electron in (as these elements are in group 1).
When these elements react they lose their outer electron. In order to lose an electron, the atom requires energy to overcome the electrostatic forces of attraction between the negatively charged electron and the positively charged nucleus.
K is the most reactive element of these 3 metals because less energy is required to remove the outer electron from its atom, that is required for sodium and lithium.
The electronic configuration will show the outer electrons.
Li 2.1
Na 2.8.1
K 2.8.8.1
The outer shell has only one electron in (as these elements are in group 1).
When these elements react they lose their outer electron. In order to lose an electron, the atom requires energy to overcome the electrostatic forces of attraction between the negatively charged electron and the positively charged nucleus.
K is the most reactive element of these 3 metals because less energy is required to remove the outer electron from its atom, that is required for sodium and lithium.
2.6 & 2.7 describe the reactions of these elements with water and understand that the reactions provide a basis for their recognition as a family of elements
Group 1 elements
Group 1 elements react vigorously with water to give an alkaline solution of the metal hydroxide as well as hydrogen gas. As we go down the group the elements are more reactive.
When these elements are reacted with water, the following observations can be seen:
Lithium
Group 1 elements react vigorously with water to give an alkaline solution of the metal hydroxide as well as hydrogen gas. As we go down the group the elements are more reactive.
When these elements are reacted with water, the following observations can be seen:
Lithium
- Moves around the surface of the water
- Hissing sound
- Bubbles of gas
- Gets smaller and smaller: eventually disappears
Sodium
- Moves around the surface of the water
- Hissing sound
- Bubbles of gas
- Melts into a shiny ball
- Gets smaller and smaller: eventually disappears
Potassium
- Moves around the surface of the water
- Hissing sound
- Bubbles of gas
- Melts into a shiny ball
- Burns with a lilac-coloured flame
- Gets smaller and smaller: eventually disappears
Trends show that the further down the group we get, the more reactive the elements are.
2.5 understand that the noble gases (Group 0) are a family of inert gases and explain their lack of reactivity in terms of their electronic configurations
The Noble Gases
Group 8/0 is called the Noble Gases. This group of gases used to be called the inert gases. Inert means chemically unreactive. These elements do not readily form compounds with other elements. Helium, although sometimes not placed in Group 8, is inert and is therefore a noble gas. The reason for the lack of reactivity is the number of electrons in the outer shell of each atom. Helium and neon atoms have full outer shells, and the rest have 8 electrons in their outer shells. None of the atoms either gains or loses electrons easily.
Group 8/0 is called the Noble Gases. This group of gases used to be called the inert gases. Inert means chemically unreactive. These elements do not readily form compounds with other elements. Helium, although sometimes not placed in Group 8, is inert and is therefore a noble gas. The reason for the lack of reactivity is the number of electrons in the outer shell of each atom. Helium and neon atoms have full outer shells, and the rest have 8 electrons in their outer shells. None of the atoms either gains or loses electrons easily.
2.4 understand why elements in the same group of the Periodic Table have similar chemical properties
Elements in the same group have similar chemical properties. This is because they have the same number of electrons in their outer shell.
2.2 &2.3 recall the positions of metals and non-metals in the Periodic Table
Metals and non-metals in the Periodic Table
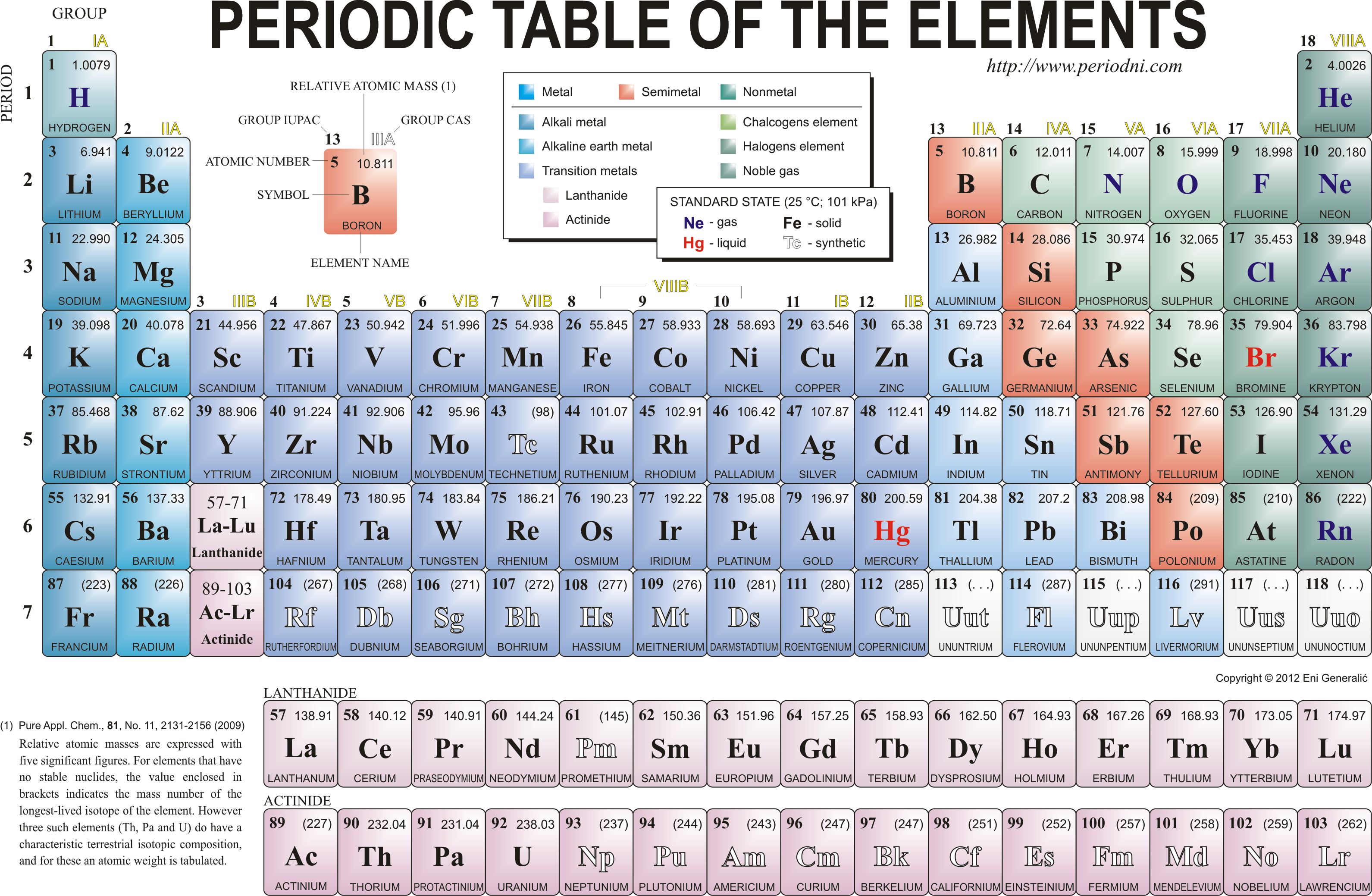
From left to right across a period there is a gradual change from metal to non-metal. For example, in Period 3, sodium, magnesium and aluminium are metals. They all conduct electricity and their oxides are basic. Phosphorus, sulfur, chlorine and argon are non-metals, They are all poor conductors of electricity and the oxides of P, S and Cl are acidic. Silicon has some properties of both a non-metal and a metal, so is therefore called a semi-metal or (metalloid). Silicon is a semi-conductor of electricity and is used in computer chips for this reason. Silicon dioxide is acidic.
2.1 understand the terms group and period
Group and period

As you can see from the periodic table of elements, it is arranged into groups and periods. Groups are the vertical groups, e.g. H, Li, Na, K, Rb, Cs and Fr. Periods are the horizontal groups, e.g. Li, Be, B, C,N, O, F, Ne.

As you can see from the periodic table of elements, it is arranged into groups and periods. Groups are the vertical groups, e.g. H, Li, Na, K, Rb, Cs and Fr. Periods are the horizontal groups, e.g. Li, Be, B, C,N, O, F, Ne.
Thursday, 16 May 2013
TRIPLE: 1.57 calculate the amounts of the products of the electrolysis of molten salts and aqueous solutions.
WILL BE COMPLETED AFTER DOUBLE EXAMS
TRIPLE: 1.56 recall that one faraday represents one mole of electrons
96500 must be the electrical charge on one mole (1 mol) of electrons (i.e. 6 x 10²³ electrons).
The value 96500 C/Mol is called the Faraday constant, after the famous scientist Michael Faraday.
The value 96500 C/Mol is called the Faraday constant, after the famous scientist Michael Faraday.
1.55 write ionic half-equations representing the reactions at the electrodes during electrolysis
Half equations
Half equations are written to show the chemical activity at the electrodes.
They need to be balanced for an exam
On the equations below, the first one is not balanced and the second has been.
Half equations are written to show the chemical activity at the electrodes.
They need to be balanced for an exam
On the equations below, the first one is not balanced and the second has been.
| Al3+ + e– → Al | Al3+ + 3e– → Al |
TRIPLE: 1.54 describe experiments to investigate electrolysis, using inert electrodes, of aqueous solutions such as sodium chloride, copper(II) sulfate and dilute sulfuric acid and predict the products
Electrolysis of aqueous sodium chloride using inert electrodes
Suitable materials for the electrodes are platinum or graphie
Particles present in solution:
Suitable materials for the electrodes are platinum or graphie
Particles present in solution:
- Sodium ions
- Chloride ions
- Water molecules
Hydrogen gas is produced as water molecules gain electrons at the cathode.
Chlorine gas is produced as chloride ions lose electrons.
1.52 understand that electrolysis involves the formation of new substances when ionic compounds conduct electricity
The products of electrolysing a molten binary compound (a compound only containing two elements) can easily be predicted by using the following rules:
- The metal is formed at the negative electrode (cathode)
- The non-metal is formed at the positive electrode (anode)
E.g. With molten lead (II) bromide, lead is formed at the negative electrode and bromine is formed at the positive electrode.
1.51 describe experiments to distinguish between electrolytes and non electrolytes
1. Set up a circuit with an LED (light emitting diode) and a break in the circuit.
2. Put both ends of the wire in a molten substance.
3. If the LED gives out light when a current is flowing, and electrolyte is present because the solution is conducting.
2. Put both ends of the wire in a molten substance.
3. If the LED gives out light when a current is flowing, and electrolyte is present because the solution is conducting.
1.50 understand why ionic compounds conduct electricity only when molten or in solution
Ionic compounds do not conduct electricity when solid since ions are not free to move.
However, ionic compounds do conduct electricity when molten or when in aqueous solution (i.e. when dissolved in water).
When an electric current is passed through a molten ionic compound, or through an aqueous solution of an ionic compound, electrolysis takes place.
However, ionic compounds do conduct electricity when molten or when in aqueous solution (i.e. when dissolved in water).
When an electric current is passed through a molten ionic compound, or through an aqueous solution of an ionic compound, electrolysis takes place.
1.49 understand why covalent compounds do not conduct electricity
In a covalent compound, there are no delocalised electrons, so it cannot hold a current. Graphite is an exception, as this is a giant covalent structure and has the 4th electron free to move, so graphite can conduct electricity.
1.48 understand that an electric current is a flow of electrons or ions
An electric current is the flow of electrons or ions because they have a charge.
1.47 explain the electrical conductivity and malleability of a metal in terms of its structure and bonding.
Most metals have high melting and boiling points because metallic bonds are strong and there are many of them to overcome in a giant structure, hence a lot of heat energy is required.
Good conductors of electricity because the delocalised electrons are free to move when a potential difference is applied across the metal.
Malleable and ductile because the layers of positive ions can easily slide over one another and take up different positions. The delocalised electrons move with them so the metallic bonds are not broken.
Good conductors of electricity because the delocalised electrons are free to move when a potential difference is applied across the metal.
Malleable and ductile because the layers of positive ions can easily slide over one another and take up different positions. The delocalised electrons move with them so the metallic bonds are not broken.
1.46 understand that a metal can be described as a giant structure of positive ions surrounded by a sea of delocalised electrons
Metallic crystals
Metals have a giant, three-dimensional lattice structure in which positive ions are arranged in a regular pattern in a 'sea of electrons'.
The outer shell electrons are detached from the atoms and are delocalised throughout the structure.
The attraction between the positive ions and the delocalised electrons is known as a metallic bond and this attraction keeps the ions together.
Metals have a giant, three-dimensional lattice structure in which positive ions are arranged in a regular pattern in a 'sea of electrons'.
The outer shell electrons are detached from the atoms and are delocalised throughout the structure.
The attraction between the positive ions and the delocalised electrons is known as a metallic bond and this attraction keeps the ions together.
TRIPLE:1.45 explain how the uses of diamond and graphite depend on their structures, limited to graphite as a lubricant and diamond in cutting
Diamond and Graphite Part II
Diamond is very hard and abrasive (strong covalent bonds that are difficult to break and it does not conduct electricity as there is no delocalised electrons. This makes it suitable to be used for cutting tools on drills because it is so abrasive. Diamond is also used for jewellery because shiny rocks seem attractive to humans.
Graphite is soft and slippery (forces of attraction between the layers are weak so the layers easily slide over one and another so can be easily separated). Graphite also conducts electricity, as there is a fourth electron that is delocalised between the layers and are free to move parallel to the layers. It is used as a lubricant because it is slippery and soft and because it is an conductor it is perfect for electrodes for electrolysis.
Diamond is very hard and abrasive (strong covalent bonds that are difficult to break and it does not conduct electricity as there is no delocalised electrons. This makes it suitable to be used for cutting tools on drills because it is so abrasive. Diamond is also used for jewellery because shiny rocks seem attractive to humans.
Graphite is soft and slippery (forces of attraction between the layers are weak so the layers easily slide over one and another so can be easily separated). Graphite also conducts electricity, as there is a fourth electron that is delocalised between the layers and are free to move parallel to the layers. It is used as a lubricant because it is slippery and soft and because it is an conductor it is perfect for electrodes for electrolysis.
TRIPLE: 1.44 draw diagrams representing the positions of the atoms in diamond and graphite
Diamond and graphite Part I

As you can see from the diagram, the diamond structure, which is made up from carbon atoms are joined to 4 other carbon atoms, this is an example of a giant covalent structure.
Graphite is another example of a giant covalent structure made out of carbon atoms. Each carbon atom is joined to 3 other carbon atom. It is an example of a lattice, where weak forces exist between each layer.

As you can see from the diagram, the diamond structure, which is made up from carbon atoms are joined to 4 other carbon atoms, this is an example of a giant covalent structure.
Graphite is another example of a giant covalent structure made out of carbon atoms. Each carbon atom is joined to 3 other carbon atom. It is an example of a lattice, where weak forces exist between each layer.
1.43 explain the high melting and boiling points of substances with giant covalent structures in terms of the breaking of many strong covalent bonds
Giant covalent structures
Some substances are made up of millions of atoms covalently bonded together to form a giant structure. These substances have high melting and boiling points, because when melting or boiling it, you are not separating inter-molecular bonds between the atoms, but the inter-molecular bonds that keep the molecule together. There are a lot of these, therefore they have high melting and boiling points.
Some substances are made up of millions of atoms covalently bonded together to form a giant structure. These substances have high melting and boiling points, because when melting or boiling it, you are not separating inter-molecular bonds between the atoms, but the inter-molecular bonds that keep the molecule together. There are a lot of these, therefore they have high melting and boiling points.
1.41 understand that substances with simple molecular structures are gases or liquids, or solids with low melting points
Simple molecular substances
Both elements and compounds can exist as simple molecular substances.
Simple molecular substances usually have low melting and boiling points.
This is because the forces of attraction between oppositely charged ions, (i.e. ionic bonds) and therefore very little energy is required to overcome them.
Although, when simple molecular substances change state, the covalent bonds between the atoms are not usually broken. Covalent bonds are strong compared to the forces of attraction between the molecules.
Both elements and compounds can exist as simple molecular substances.
Simple molecular substances usually have low melting and boiling points.
This is because the forces of attraction between oppositely charged ions, (i.e. ionic bonds) and therefore very little energy is required to overcome them.
Although, when simple molecular substances change state, the covalent bonds between the atoms are not usually broken. Covalent bonds are strong compared to the forces of attraction between the molecules.
1.40 explain, using dot and cross diagrams, the formation of covalent compounds by electron sharing for the following substances:
i hydrogen
ii chlorine
iii hydrogen chloride
iv water
 Just one example of a dot cross diagram.
Just one example of a dot cross diagram.v methane
vi ammonia
vii oxygen
viii nitrogen
ix carbon dioxide
x ethane
xi ethene
1.39 understand covalent bonding as a strong attraction between the bonding pair of electrons and the nuclei of the atoms involved in the bond
Covalent bonding part II
During covalent bonding, the sharing of electrons between 2 non-metals, the electrons become attracted to the nucleus of each ion in a bond, this is electrostatic attraction.
During covalent bonding, the sharing of electrons between 2 non-metals, the electrons become attracted to the nucleus of each ion in a bond, this is electrostatic attraction.
1.38 describe the formation of a covalent bond by the sharing of a pair of electrons between two atoms
Covalent bonds
When two non-metallic elements combine together they often share electrons between them. When an atom of hydrogen, H and an atom of chlorine, Cl, combine to form hydrogen chloride, HCl, the atoms come close enough together for their outer electron (valence) shells to overlap.
When two non-metallic elements combine together they often share electrons between them. When an atom of hydrogen, H and an atom of chlorine, Cl, combine to form hydrogen chloride, HCl, the atoms come close enough together for their outer electron (valence) shells to overlap.
TRIPLE: 1.36 &1.37 describe an ionic crystal as a giant three-dimensional lattice structure held together by the attraction between oppositely charged ions
Giant ionic lattice structure
As you can see from the diagram, this is a giant ionic lattice. When an ionic compound forms, the positively charged ions attract the negatively charged ions and arrange themselves into a three-dimensional structure called an ionic lattice. The arrangement of the ions in the ionic lattice of sodium chloride is shown above.
As you can see from the diagram, this is a giant ionic lattice. When an ionic compound forms, the positively charged ions attract the negatively charged ions and arrange themselves into a three-dimensional structure called an ionic lattice. The arrangement of the ions in the ionic lattice of sodium chloride is shown above.
TRIPLE: 1.35 understand the relationship between ionic charge and the melting point and boiling point of an ionic compound
The relationship between the ionic charge and boiling/melting points
For ions of similar size, the strength of the forces of attraction between the ions will depend on the size of their charge. For this reason, magnesium oxide (Mg²+O²-) has a higher melting and boiling point than sodium chloride (Na+Cl-). The more electrons that have been transferred, the stronger the bond because of the stronger electrostatic forces.
For ions of similar size, the strength of the forces of attraction between the ions will depend on the size of their charge. For this reason, magnesium oxide (Mg²+O²-) has a higher melting and boiling point than sodium chloride (Na+Cl-). The more electrons that have been transferred, the stronger the bond because of the stronger electrostatic forces.
1.33 & 1.34 understand ionic bonding as a strong electrostatic attraction between oppositely charged ions
Electrostatic attraction in ionic bonding
The electrostatic attraction keeps the ions bonded, and as it is very strong, it takes a lot of energy to break these bonds.
For this reason, ionic compounds have very high melting and boiling points, because of the amount of energy needed to break these strong bonds between oppositely charged ions.
The electrostatic attraction keeps the ions bonded, and as it is very strong, it takes a lot of energy to break these bonds.
For this reason, ionic compounds have very high melting and boiling points, because of the amount of energy needed to break these strong bonds between oppositely charged ions.
1.32 explain, using dot and cross diagrams, the formation of ionic compounds by electron transfer, limited to combinations of elements from Groups 1, 2, 3 and 5, 6, 7
Dot and cross diagrams
Dot and cross diagrams are used to show electron transfers, and the creation of ions.
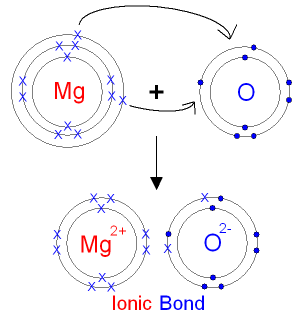
As you can see, the magnesium atom gives 2 electrons to the oxygen atom, so an ionic bond is created and therefore, 2 ions are created; the magnesium atom becomes a positive ion, losing 2 electrons and the oxygen atom becomes a negative ion, gaining 2 electrons.
Dot and cross diagrams are used to show electron transfers, and the creation of ions.

As you can see, the magnesium atom gives 2 electrons to the oxygen atom, so an ionic bond is created and therefore, 2 ions are created; the magnesium atom becomes a positive ion, losing 2 electrons and the oxygen atom becomes a negative ion, gaining 2 electrons.
1.31 deduce the charge of an ion from the electronic configuration of the atom from which the ion is formed
Electronic configuration of ions
Na has the electronic configuration of 2.8.1
Its positive ion, Na+ has the electronic configuration of 2.8
Cl has the electronic configuration of 2.8.7
Its negative ion, Cl- has the electronic configuration of 2.8.8
Na has the electronic configuration of 2.8.1
Its positive ion, Na+ has the electronic configuration of 2.8
Cl has the electronic configuration of 2.8.7
Its negative ion, Cl- has the electronic configuration of 2.8.8
1.30 recall the charges of common ions in this specification
Common ions
There are loads, mainly positive but also negative ions too.
Positive ions....
There are loads, mainly positive but also negative ions too.
Positive ions....
- Lithium-Li+
- Sodium- Na+
- Potassium- K+
- Silver-Ag+
- Copper (I)-Cu+
- Ammonium-NH₄+
- Magnesium-Mg ²+
- Calcium- Ca ²+
- Barium- Ba²+
- Zinc- Zn ²+
- Copper (II)-Cu ²+
- Lead (II)- Pb²+
- Iron (II)-Fe²+
- Aluminum- Al 3+
- Chromium (III) 3+
- Lead (IV) 4+
- Maganase (IV)4+
Negative ions
- Fluoride - Fl-
- Chloride- Cl-
- Bromide- Br-
- Iodide- I-
- Hydroxide- OH-
- Nitrate- NO3-
- Oxide- O²-
- Sulfide- S²-
- Sulfate-SO4²-
- Carbonate CO₃ ²-
1.29 understand oxidation as the loss of electrons and reduction as the gain of electrons
Oxidation and reduction
Oxidation is the process of an atom losing an electron/s and becoming an positively charged ion.
Reduction is the process of an atom gaining an electron/s and becoming an negatively charged ion.
You can remember this using OIL RIG.
OXIDATION IS LOSING
REDUCTION IS GAINING
Oxidation is the process of an atom losing an electron/s and becoming an positively charged ion.
Reduction is the process of an atom gaining an electron/s and becoming an negatively charged ion.
You can remember this using OIL RIG.
OXIDATION IS LOSING
REDUCTION IS GAINING
1.28 describe the formation of ions by the gain or loss of electrons
The formation of ions
An ion is an electrically charged atom or group of atoms. Ions are formed by the loss or gain of electrons.
If an atom gains an electron it becomes a negatively charged ion.
If an atom loses an electron it becomes a positively charged ion.
An ion is an electrically charged atom or group of atoms. Ions are formed by the loss or gain of electrons.
If an atom gains an electron it becomes a negatively charged ion.
If an atom loses an electron it becomes a positively charged ion.
1.27 carry out mole calculations using volumes and molar concentrations
Calculating volumes and molar concentrations
Calculate the volume of gas (carbon dioxide) produced when 50g of calcium carbonate is decomposed by heating
1. Calculate the amount, in moles, of calcium carbonate reacted
Mr of CaCO₃ is 100
Amount of CaCO₃ = (50 ÷ 100) = 0.5 mol
2. Calculate the amount, in moles, of carbon dioxide formed
CaCO₃ --> CaO + CO₂
1 mol of CaCO₃ produces 1 mol of CO₂
0.5 mol of CaCO₃ produces 0.5 mol of CO₂
3. Calculate the volume of CO₂ formed
Volume of CO₂ = (0.5 x 24) dm³ = 12dm³
Calculate the volume of gas (carbon dioxide) produced when 50g of calcium carbonate is decomposed by heating
1. Calculate the amount, in moles, of calcium carbonate reacted
Mr of CaCO₃ is 100
Amount of CaCO₃ = (50 ÷ 100) = 0.5 mol
2. Calculate the amount, in moles, of carbon dioxide formed
CaCO₃ --> CaO + CO₂
1 mol of CaCO₃ produces 1 mol of CO₂
0.5 mol of CaCO₃ produces 0.5 mol of CO₂
3. Calculate the volume of CO₂ formed
Volume of CO₂ = (0.5 x 24) dm³ = 12dm³
TRIPLE: 1.26 calculate percentage yield
Percentage yield
Often in a chemical reaction, the theoretical yield (the maximum amount of a product that could be formed given the amount of the reactant) is not obtained.
For this reason, it is useful to calculate the percentage yield to find out the efficiency of the reaction.
Percentage yield= yield obtained/theoretical yield x 100
Exam Question Example:
In an experiment to displace copper from copper sulfate, 6.5g of Zinc was added to an excess of copper (II) sulfate solution. The copper was filtered off, washed and dried. The mass of copper obtained was 4.8g.
Calculate the percentage yield of copper
The equation for the reaction is Zn(s) + CuSo₄(aq) ---> ZnSo₄ (aq) + Cu (s)
1. Calculate the amount, in moles, of zinc reacted
Amount of zinc = 6.5/65 = 0.10mol
2. Calculate the maximum amount of copper that could be formed
Maximum amount of copper = 0.10 mol
3. Calculate the maximum mass of copper that could be formed
Maximum mass of copper = (0.10 x 64)= 6.4g
4. Calculate percentage yield of copper
Percentage yield = 4.8/6.4 x 100 = 75%
Often in a chemical reaction, the theoretical yield (the maximum amount of a product that could be formed given the amount of the reactant) is not obtained.
For this reason, it is useful to calculate the percentage yield to find out the efficiency of the reaction.
Percentage yield= yield obtained/theoretical yield x 100
Exam Question Example:
In an experiment to displace copper from copper sulfate, 6.5g of Zinc was added to an excess of copper (II) sulfate solution. The copper was filtered off, washed and dried. The mass of copper obtained was 4.8g.
Calculate the percentage yield of copper
The equation for the reaction is Zn(s) + CuSo₄(aq) ---> ZnSo₄ (aq) + Cu (s)
1. Calculate the amount, in moles, of zinc reacted
Amount of zinc = 6.5/65 = 0.10mol
2. Calculate the maximum amount of copper that could be formed
Maximum amount of copper = 0.10 mol
3. Calculate the maximum mass of copper that could be formed
Maximum mass of copper = (0.10 x 64)= 6.4g
4. Calculate percentage yield of copper
Percentage yield = 4.8/6.4 x 100 = 75%
1.25 calculate reacting masses using experimental data and chemical equations
Reacting masses
Typical exam question:
Calculate the mass of magnesium oxide that can be made by completely burning 6g of magnesium in oxygen.
Equation for reaction: 2Mg + O₂----> 2MgO
1. Calculate the amount, in moles, of magnesium reacted
Ar of Mg is 24
Amount of magnesium = (6 ÷ 24) = 0.25mol
2. Calculate the amount of magnesium oxide formed
The equation tells us that 2 mol of Mg for 2 mol of MgO, hence the amount of MgO formed is the same as the amount of Mg reacted.
Amount of MgO formed is 0.25 mol
3. Calculate the mass of MgO formed
Mr of MgO + (24 + 16) = 40
Mass of magnesium oxide = (0.25 x 40) = 10g
Typical exam question:
Calculate the mass of magnesium oxide that can be made by completely burning 6g of magnesium in oxygen.
Equation for reaction: 2Mg + O₂----> 2MgO
1. Calculate the amount, in moles, of magnesium reacted
Ar of Mg is 24
Amount of magnesium = (6 ÷ 24) = 0.25mol
2. Calculate the amount of magnesium oxide formed
The equation tells us that 2 mol of Mg for 2 mol of MgO, hence the amount of MgO formed is the same as the amount of Mg reacted.
Amount of MgO formed is 0.25 mol
3. Calculate the mass of MgO formed
Mr of MgO + (24 + 16) = 40
Mass of magnesium oxide = (0.25 x 40) = 10g
Wednesday, 15 May 2013
1.24 calculate empirical and molecular formulae from experimental data
Empirical Formula
The empirical formula of a compound gives the simplest whole-number ratio of atoms of each element in a compound.
It can be calculated from knowledge of the ratio of masses of each element in the compound.
e.g. A compound that contains 10g of hydrogen and 80g of oxygen has an empirical formula of H₂O.
This can be shown here:
Amount of hydrogen atoms= mass in grams ÷ Ar of hydrogen = (10 ÷ 1)= 10 mol
Amount of oxygen atoms= mass in grams ÷ Ar of oxygen = (80÷ 16) = 5 mol
Therefore the formula/ratio is 10:5 or simplified to be 2:1
The empirical formula of a compound gives the simplest whole-number ratio of atoms of each element in a compound.
It can be calculated from knowledge of the ratio of masses of each element in the compound.
e.g. A compound that contains 10g of hydrogen and 80g of oxygen has an empirical formula of H₂O.
This can be shown here:
Amount of hydrogen atoms= mass in grams ÷ Ar of hydrogen = (10 ÷ 1)= 10 mol
Amount of oxygen atoms= mass in grams ÷ Ar of oxygen = (80÷ 16) = 5 mol
Therefore the formula/ratio is 10:5 or simplified to be 2:1
1.23 understand how the formulae of simple compounds can be obtained experimentally, including metal oxides, water and salts containing water of crystallisation
Formula of simple compounds
To find this you could conduct an experiment:
1. Weigh the compound
2. Destroy the bonds and separate the mixture through crystallisation
3. Weigh the different substances that have been left
4. Deduce the measurements for the formula
To find this you could conduct an experiment:
1. Weigh the compound
2. Destroy the bonds and separate the mixture through crystallisation
3. Weigh the different substances that have been left
4. Deduce the measurements for the formula
1.22 use the state symbols (s), (l), (g) and (aq) in chemical equations to represent solids, liquids, gases and aqueous solutions respectively
State Symbols
State symbols are sometimes written after formulae in chemical equations to show which physical state each substance is in.
There are four different state symbols that you need to know:
(s)- Solid
(l)- Liquid
(g)- Gas
(aq)- Aqueous
State symbols are very useful, because they tell you the conditions required for a reaction to take place.
State symbols are sometimes written after formulae in chemical equations to show which physical state each substance is in.
There are four different state symbols that you need to know:
(s)- Solid
(l)- Liquid
(g)- Gas
(aq)- Aqueous
State symbols are very useful, because they tell you the conditions required for a reaction to take place.
1.21 write word equations and balanced chemical equations to represent the reactions studied in this specification
Writing balanced chemical equations
There has to be the same number of each atoms/element on both sides of the equation.
Aluminum + copper (II) oxide----> Aluminum oxide + copper
Al + CuO ----> Al₂O₃ + Cu
1 Al 2Al
2 Cu 1Cu
1 O 3 O
There needs to be the same amount of each elements on both sides, so we must balance it.
2Al + 3CuO ----> Al₂O₃ + 3Cu
2 Al 2Al
3Cu 3Cu
3 O 3O
Now the equation is balanced.
There has to be the same number of each atoms/element on both sides of the equation.
Aluminum + copper (II) oxide----> Aluminum oxide + copper
Al + CuO ----> Al₂O₃ + Cu
1 Al 2Al
2 Cu 1Cu
1 O 3 O
There needs to be the same amount of each elements on both sides, so we must balance it.
2Al + 3CuO ----> Al₂O₃ + 3Cu
2 Al 2Al
3Cu 3Cu
3 O 3O
Now the equation is balanced.
TRIPLE: 1.20 understand the term molar volume of a gas and use its values (24 dm3 and 24,000 cm3 ) at room temperature and pressure (rtp) in calculations.
Molar volume of a gas
One mole of any gas has a volume of 24dm³
at room temperature and pressure, this is called the molar volume of a gas.
volume of gas (in dm³) = amount x 24
If we had 3 mols of Hydrogen, the volume of the gas would be
(3 x 24) = 72dm³
1.19 carry out mole calculations using relative atomic mass (Ar ) and relative formula mass (Mr )
Mole calculations
The mass of one mole of atoms is easily calculated, it is simply the relative atomic mass expressed in grams.
Hydrogen- Ar=1 so 1g
Carbon- Ar= 12 so 12g
The mass of one mole of a substance is also easily calculated by working out the formula mass (Mr) and expressing it in grams:
H₂ - Mr = 2 so mole is 2g
The mass of one mole of atoms is easily calculated, it is simply the relative atomic mass expressed in grams.
Hydrogen- Ar=1 so 1g
Carbon- Ar= 12 so 12g
The mass of one mole of a substance is also easily calculated by working out the formula mass (Mr) and expressing it in grams:
H₂ - Mr = 2 so mole is 2g
1.17 + 1.18 understand the use of the term mole to represent the amount of substance and know the term 'Avogadro Number'
Moles
The mole is a measure of the amount of substance.
The mole (1 mol) is the amount of substance that contains:
6 x 1023 particles of the substance.
This is known as the Avogadro Number.
e.g.
1 mol of Sodium (Na) contains 6 x 1023 atoms of sodium.
The mole is a measure of the amount of substance.
The mole (1 mol) is the amount of substance that contains:
6 x 1023 particles of the substance.
This is known as the Avogadro Number.
e.g.
1 mol of Sodium (Na) contains 6 x 1023 atoms of sodium.
1.16 calculate relative formula masses (Mr ) from relative atomic masses (Ar)
Relative formula masses
Relative formula mass is given the symbol, Mr.
To calculate the Mr, of a substance, all you have to do is add up the relative atomic masses of all the atoms present in the formula.
Water- H₂0
Atoms present:(2 x H) + (1 x O)
Mr= (2 x 1) + 16= 18
There is 1 electron per hydrogen atom, and 2 hydrogen atoms, so that is 2 electrons. There is also 1 oxygen atom, which has 16 electrons, 2 + 16= 18
Relative formula mass is given the symbol, Mr.
To calculate the Mr, of a substance, all you have to do is add up the relative atomic masses of all the atoms present in the formula.
Water- H₂0
Atoms present:(2 x H) + (1 x O)
Mr= (2 x 1) + 16= 18
There is 1 electron per hydrogen atom, and 2 hydrogen atoms, so that is 2 electrons. There is also 1 oxygen atom, which has 16 electrons, 2 + 16= 18
Atomic Structure 1.15 deduce the number of outer electrons in a main group element from its position in the Periodic Table.
Number of electrons in groups of the Periodic Table
As you can see, some of the columns of the table have numbers above them, this tells us how many electrons are in the outer shell of that group. It is a good idea to label this on your periodic table in your chemistry exam.
As you can see, some of the columns of the table have numbers above them, this tells us how many electrons are in the outer shell of that group. It is a good idea to label this on your periodic table in your chemistry exam.
Atomic Structure: 1.14 deduce the electronic configurations of the first 20 elements from their positions in the Periodic Table
Electronic configuration
The arrangement of electrons into shells for an atom is known as its electronic configuration
As you are given a periodic table in the exam, you don't have to remember all of them, just be able to state them when asked.
They are given in shorthand notations, e.g.
Carbon has 6 electrons, so its EC would be 2.4
Magnesium has 12 electrons, so its EC would be 2.8.2
The arrangement of electrons into shells for an atom is known as its electronic configuration
As you are given a periodic table in the exam, you don't have to remember all of them, just be able to state them when asked.
They are given in shorthand notations, e.g.
Carbon has 6 electrons, so its EC would be 2.4
Magnesium has 12 electrons, so its EC would be 2.8.2
Atomic Structure 1.13 understand that the Periodic Table is an arrangement of elements in order of atomic number
Periodic Table
As you can see the periodic table, shows all the elements known to man, and is arranged in periods and groups. It shows the arrangement of elements in order of atomic number.
Atomic Structure 1.12 calculate the relative atomic mass of an element from the relative abundances of its isotopes
Calculating the relative atomic mass
Example:
Chlorine has two isotopes: chlorine-35 and chlorine-37
A typical sample of chlorine will be 75% chlorine-35 atoms and 25% chlorine-37 atoms.
Total mass of 100 atoms= (75 x 35) + 5 x 37)= 3550
Mean mass of 1 atom= (3550 ÷ 100)= 35.5
Ar of chlorine is 35.5
Example:
Chlorine has two isotopes: chlorine-35 and chlorine-37
A typical sample of chlorine will be 75% chlorine-35 atoms and 25% chlorine-37 atoms.
Total mass of 100 atoms= (75 x 35) + 5 x 37)= 3550
Mean mass of 1 atom= (3550 ÷ 100)= 35.5
Ar of chlorine is 35.5
Atomic Structure 1.11 understand the terms atomic number, mass number, isotopes and relative atomic mass (Ar )
Atomic number, mass number, isotopes and relative atomic mass
 Atomic number= number of protons in the nucleus of an atom
Atomic number= number of protons in the nucleus of an atom
Mass number= number of protons + number of neutrons in the nucleus of an atom
Isotopes are atoms that have the same atomic number but different mass numbers. This is because they contain the same number of protons but different numbers of neutrons.
Relative atomic mass is calculated from the masses and relative abundances of all the isotopes of a particular element. It is usually given in the symbol Ar.
Mass number= number of protons + number of neutrons in the nucleus of an atom
Isotopes are atoms that have the same atomic number but different mass numbers. This is because they contain the same number of protons but different numbers of neutrons.
Relative atomic mass is calculated from the masses and relative abundances of all the isotopes of a particular element. It is usually given in the symbol Ar.
Atomic Structure 1.10 recall the relative mass and relative charge of a proton, neutron and electron
Relative mass and relative charge
Neutron:
Neutron:
Relative charge: 0
Relative mass: 1
Proton:
Relative charge: +1
Relative mass: 1
Electron:
Relative charge: -1
Relative mass: 1/1836
Nearly all the mass is concentrated in the nucleus.
Subscribe to:
Comments (Atom)